The Periodic Table
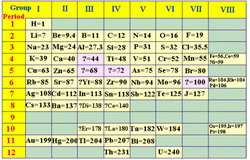
In 1869, a Russian chemistry by the name of Dmitri Mendeleev produced the first orderly arrangement of the discovered elements. At that time, Mendeleev only knew of 63 discovered elements. The image to the left is an example of what Mendeleev's early periodic table looked like. When Mendeleev was organizing the elements, he arranged them in order of increasing atomic mass and he took into consideration the chemical properties of the element. If he noticed that the chemical properties of the elements repeated, he placed them into rows with each other.
There are two key things to observe about Mendeleev's periodic table: periodicity and gaps. Today, we refer to Mendeleev's organization material relating to repeating chemical properties as periodicity, which connects us to the modern day periodic law. The periodic law states that when our 118 known elements are arranged according to atomic number in the modern periodic table, specific properties will appear in the elements at regular intervals. The second observation about Mendeleev's periodic table was that he left gaps in his rows and columns as to predict the placement of elements that had yet to be discovered. For example, in the image to the left, find the gap with a ? = 72... that gap represented where Germanium (atomic number 32) now fits into the periodic table.
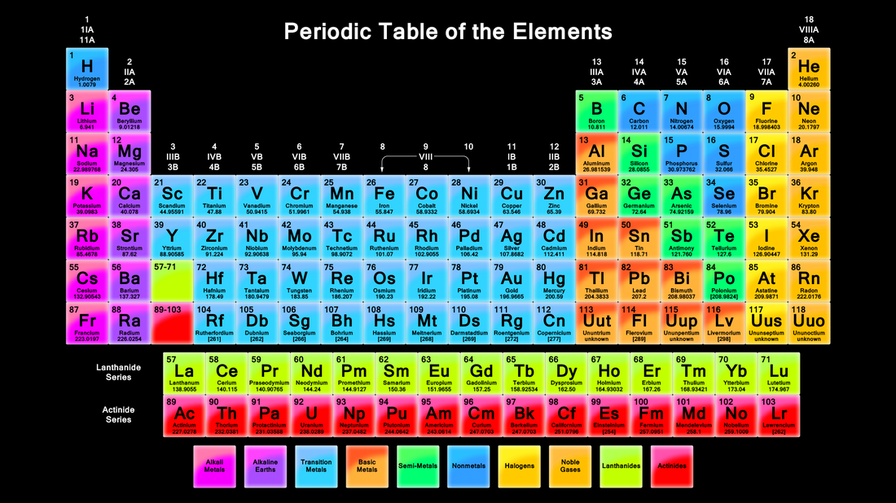
The modern periodic table includes 118 elements and three major sections: metals, nonmetal, and metalloids. Within the metals, nonmetals, and metalloids sections, there are individual subsections based on the different properties of those elements within those areas of the periodic table. Watch the Voicethread below to learn more about the periodic table and how to understand the set up.
In 1869, a Russian chemistry by the name of Dmitri Mendeleev produced the first orderly arrangement of the discovered elements. At that time, Mendeleev only knew of 63 discovered elements. The image to the left is an example of what Mendeleev's early periodic table looked like. When Mendeleev was organizing the elements, he arranged them in order of increasing atomic mass and he took into consideration the chemical properties of the element. If he noticed that the chemical properties of the elements repeated, he placed them into rows with each other.
There are two key things to observe about Mendeleev's periodic table: periodicity and gaps. Today, we refer to Mendeleev's organization material relating to repeating chemical properties as periodicity, which connects us to the modern day periodic law. The periodic law states that when our 118 known elements are arranged according to atomic number in the modern periodic table, specific properties will appear in the elements at regular intervals. The second observation about Mendeleev's periodic table was that he left gaps in his rows and columns as to predict the placement of elements that had yet to be discovered. For example, in the image to the left, find the gap with a ? = 72... that gap represented where Germanium (atomic number 32) now fits into the periodic table.
The modern periodic table includes 118 elements and three major sections: metals, nonmetal, and metalloids. Within the metals, nonmetals, and metalloids sections, there are individual subsections based on the different properties of those elements within those areas of the periodic table. Watch the Voicethread below to learn more about the periodic table and how to understand the set up.
VoiceThread is also available at https://voicethread.com/share/4774263/.
Meet the elements and explore their uses by watching the Meet the Elements - They Might Be Giants video below. While you watch the video, write down any of the elements and their uses that surprised you!
Practice Problems
|
| ||||||||||||