Matter: Changes and Reactions
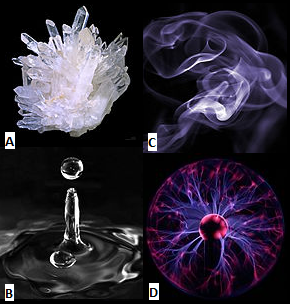
In chemistry, the most basic definition of matter is anything that has a mass and takes up space or has a volume. Matter has four primary forms or states: solid, liquid, gas, and plasma. Solid, liquid, and gas are more commonly referred to but plasma is gradually becoming a more understood state of matter. Depending on the temperature and pressure, matter can transition between the phases of matter. When the phase transition happens, that substance of matter is undergoing physical changes. In addition to the phase transitions, matter can and will undergo chemical changes based on interactions with the environment. Those chemical changes occur during different types of chemical reactions. There is one major law that connects with the physical and chemical changes that matter experiences except for nuclear matter and reactions. The law is known as the law of conservation of mass and it states that mass can neither be created nor deserved, however the mass can be rearranged or change in form.
In chemistry, the most basic definition of matter is anything that has a mass and takes up space or has a volume. Matter has four primary forms or states: solid, liquid, gas, and plasma. Solid, liquid, and gas are more commonly referred to but plasma is gradually becoming a more understood state of matter. Depending on the temperature and pressure, matter can transition between the phases of matter. When the phase transition happens, that substance of matter is undergoing physical changes. In addition to the phase transitions, matter can and will undergo chemical changes based on interactions with the environment. Those chemical changes occur during different types of chemical reactions. There is one major law that connects with the physical and chemical changes that matter experiences except for nuclear matter and reactions. The law is known as the law of conservation of mass and it states that mass can neither be created nor deserved, however the mass can be rearranged or change in form.
Physical and Chemical Changes
All around us physical and chemical changes are occurring in matter and substances. A physical change in matter does not change what the substance is or the chemical composition of the substance. An example of a physical change would be taking a piece of paper and cutting the paper into smaller pieces or freezing water. When matter is undergoing chemical change, a new substance is formed and energy is either absorbed (endothermic reactions - heat energy is being absorbed into the substance) or given off (exothermic reactions - heat energy is being released from the substance). The speed of the chemical reactions depends on temperature, pressure, and concentrations of the substances involved. An example of a chemical change would be digesting food or burning wood.
All around us physical and chemical changes are occurring in matter and substances. A physical change in matter does not change what the substance is or the chemical composition of the substance. An example of a physical change would be taking a piece of paper and cutting the paper into smaller pieces or freezing water. When matter is undergoing chemical change, a new substance is formed and energy is either absorbed (endothermic reactions - heat energy is being absorbed into the substance) or given off (exothermic reactions - heat energy is being released from the substance). The speed of the chemical reactions depends on temperature, pressure, and concentrations of the substances involved. An example of a chemical change would be digesting food or burning wood.
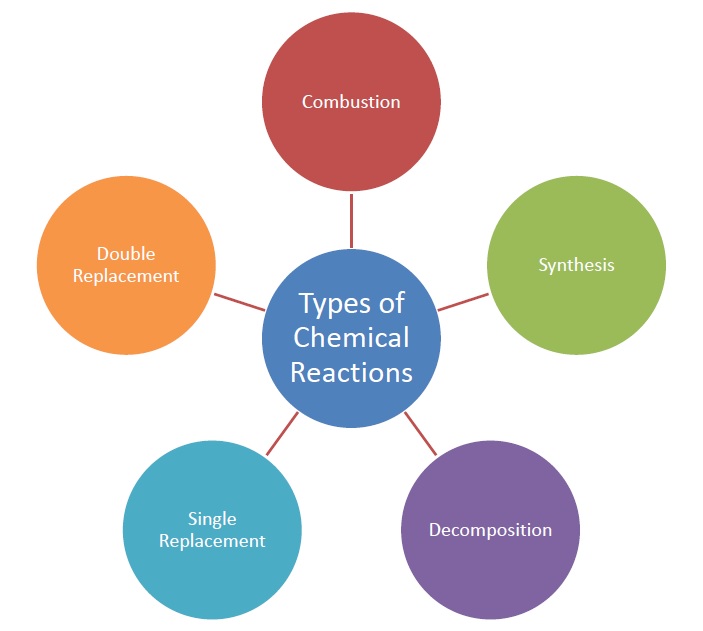
Types of Chemical Reactions
|
VoiceThread also available at https://voicethread.com/share/4782749/.
|
| ||||||
Assigned Practice Problems
|
| ||||||||||||||||||