About the Atom
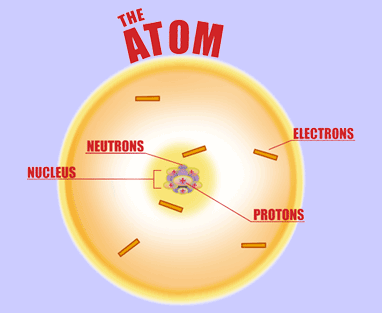
Atoms are all around us and atoms are often referred to as the building blocks of life. Every atom is made up of three extremely tiny subatomic particles: protons, electrons, and neutrons. The protons and neutrons make up the nucleus which is located in the center of the atom while the electrons surround the nucleus in the electron cloud of the atom.
The electron has a negative charge of 1.6022 x 10^-19 Coulomb and a mass of 0.000548591 a.m.u. or 9.1 x 10^-31 kg. The electron is often represented with the symbol "e" and is the smallest subatomic particle.
The proton has a positive charge of 1.6022 x 10^-19 Coulomb and a mass of 1.0072766 a.m.u. or 1.6726 x 10^-27 kg. The proton is symbolically represented with the letter "p" and is approximately 1837 times heavier than the electron.
The electron and proton have opposite charges but the magnitudes of the charges are identical. Due to the opposite charges, the protons and electrons attract to one another and hold our atom together.
The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The neutron is found in the nucleus of the atom with the proton. The mass of the neutron is 1.0086654 a.m.u. or 1.6749 x 10^-27 kg, making it the largest subatomic particle. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.
In the video below, Bill Nye explores the concept of the atom.
Atoms are all around us and atoms are often referred to as the building blocks of life. Every atom is made up of three extremely tiny subatomic particles: protons, electrons, and neutrons. The protons and neutrons make up the nucleus which is located in the center of the atom while the electrons surround the nucleus in the electron cloud of the atom.
The electron has a negative charge of 1.6022 x 10^-19 Coulomb and a mass of 0.000548591 a.m.u. or 9.1 x 10^-31 kg. The electron is often represented with the symbol "e" and is the smallest subatomic particle.
The proton has a positive charge of 1.6022 x 10^-19 Coulomb and a mass of 1.0072766 a.m.u. or 1.6726 x 10^-27 kg. The proton is symbolically represented with the letter "p" and is approximately 1837 times heavier than the electron.
The electron and proton have opposite charges but the magnitudes of the charges are identical. Due to the opposite charges, the protons and electrons attract to one another and hold our atom together.
The neutron differs from the proton and electron because it is a neutral atom, meaning it has no charge. The neutron is found in the nucleus of the atom with the proton. The mass of the neutron is 1.0086654 a.m.u. or 1.6749 x 10^-27 kg, making it the largest subatomic particle. The neutron is only slightly larger than the proton but it is 1842 times heavier than the electron.
In the video below, Bill Nye explores the concept of the atom.
Understanding the History of the Atomic Models
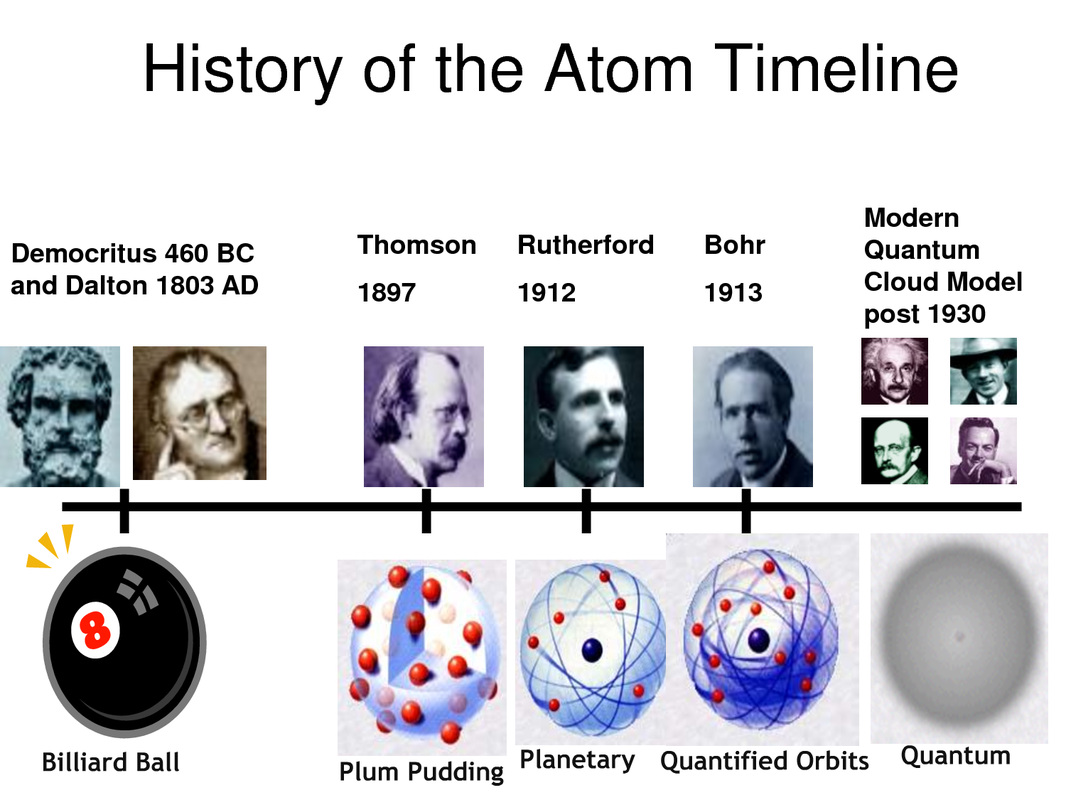
Our understanding of the atom has changed drastically over the years. There are five primary models of the atoms that different scientists developed over the years. In 460 BC, Democritus developed his theory of the atom, which was that everything is composed of atoms and that the atoms are composed of empty space but are not destructible. Democritus' belief led to the development of the Billiard Ball model in 1803 AD by Dalton. Dalton's model three main components: atoms are the smallest part of elements, different elements have different atoms with different respective masses, and atoms are solid spheres that are indestructible, which led to the name of the billiard ball model.
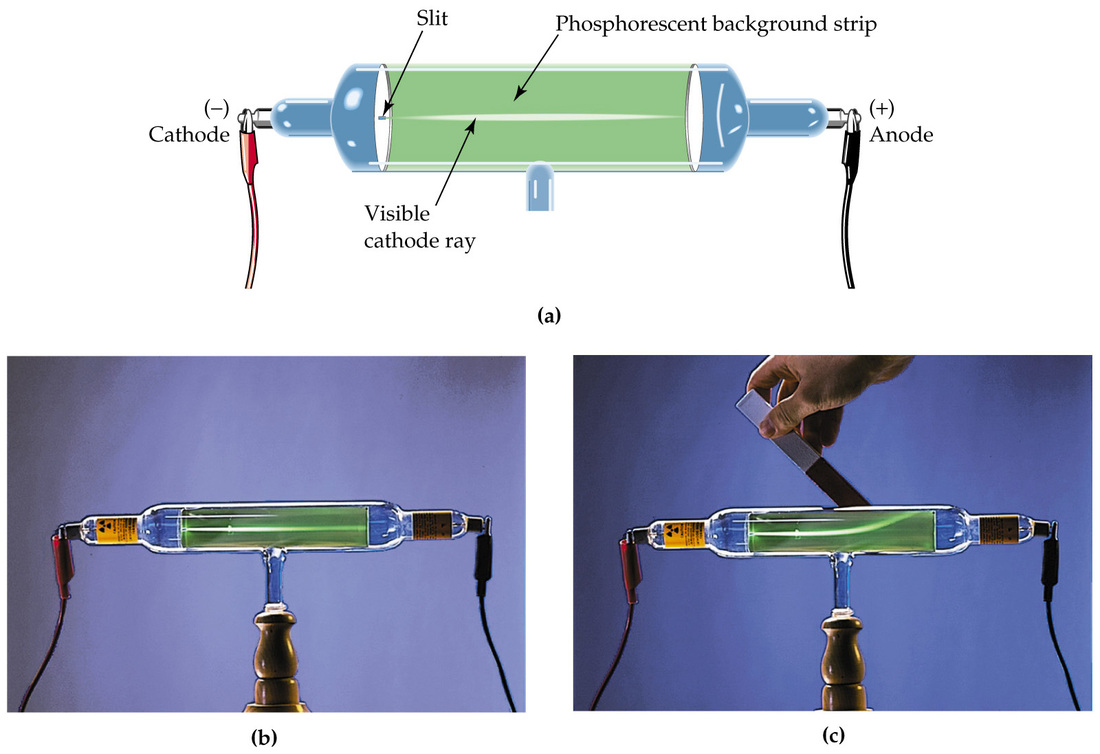
In 1897, J.J. Thomson proposed his plum pudding model for the atom after experimenting with the cathode ray tube. J.J. Thomson discovered the electron and developed his atomic model around the electron. In his model, the atom is filled with negatively charged electrons (plums) surrounded by a positive charge (pudding).
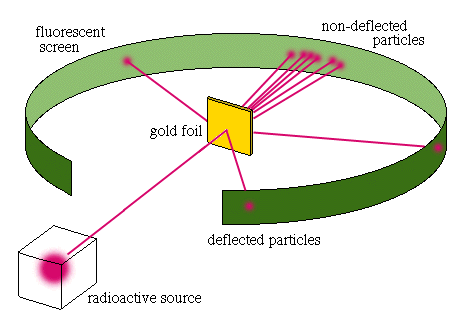
The next model was created by Ernest Rutherford in 1912. Rutherford conducted his Gold Foil Experiment and found that the atom had a relatively high concentrated charge located toward the center of the atom. The high area of concentrated charge (protons) was found to be small in volume in comparison to the rest of the atom. His planetary model included a new feature of the modern day nucleus (sun) located at the center of the atom surrounded by negatively charged electrons (orbiting planets).
In 1913, Niels Bohr developed the quantified orbits model of the atom. Bohr's model was very similar to Ernest Rutherford's Planetary model but Bohr focused on a quantum physical interpretation of the movement of the electrons. For Bohr, the electrons still traveled around a small, positively charged nucleus but the movement connected to electrostatic forces as well as specific quantum levels of energy.
The modern day - quantum cloud model was developed after 1930. In the quantum cloud model, the electrons are no longer traveling around the nucleus in a fixed orbit but the atoms remain very small in size. The model recognizes the quantum mechanically influences in an atom and states that we can not know for certain where the electrons are located so we can describe their probable location around the positively charged, central nucleus using a cloud depiction.
Our current understanding of the atom as well as the protons, neutrons, and electrons directly connects with our periodic table of elements. The atomic number of the elements connects to the number of protons and electrons in the atoms of that element. The amount of protons will always equal the amount of electrons in an atom, the equal but opposite charge of the protons and electrons hold the atom together (electrostatic forces). While the atom is held together due to oppositely charged particles attracting one another, there are also repulsion forces acting between similarly charged particles in the atom (proton to proton or electron to electron interaction). In order to find the number of neutrons in the atoms of a particular element, we must round the atomic mass to the nearest whole number and subtract the atomic number. Found in the nucleus of the atom, the neutrons are neutral in charge so those particles are not influenced by the positive charge of the proton or the negative charge of the electrons.
Our understanding of the atom has changed drastically over the years. There are five primary models of the atoms that different scientists developed over the years. In 460 BC, Democritus developed his theory of the atom, which was that everything is composed of atoms and that the atoms are composed of empty space but are not destructible. Democritus' belief led to the development of the Billiard Ball model in 1803 AD by Dalton. Dalton's model three main components: atoms are the smallest part of elements, different elements have different atoms with different respective masses, and atoms are solid spheres that are indestructible, which led to the name of the billiard ball model.
In 1897, J.J. Thomson proposed his plum pudding model for the atom after experimenting with the cathode ray tube. J.J. Thomson discovered the electron and developed his atomic model around the electron. In his model, the atom is filled with negatively charged electrons (plums) surrounded by a positive charge (pudding).
The next model was created by Ernest Rutherford in 1912. Rutherford conducted his Gold Foil Experiment and found that the atom had a relatively high concentrated charge located toward the center of the atom. The high area of concentrated charge (protons) was found to be small in volume in comparison to the rest of the atom. His planetary model included a new feature of the modern day nucleus (sun) located at the center of the atom surrounded by negatively charged electrons (orbiting planets).
In 1913, Niels Bohr developed the quantified orbits model of the atom. Bohr's model was very similar to Ernest Rutherford's Planetary model but Bohr focused on a quantum physical interpretation of the movement of the electrons. For Bohr, the electrons still traveled around a small, positively charged nucleus but the movement connected to electrostatic forces as well as specific quantum levels of energy.
The modern day - quantum cloud model was developed after 1930. In the quantum cloud model, the electrons are no longer traveling around the nucleus in a fixed orbit but the atoms remain very small in size. The model recognizes the quantum mechanically influences in an atom and states that we can not know for certain where the electrons are located so we can describe their probable location around the positively charged, central nucleus using a cloud depiction.
Our current understanding of the atom as well as the protons, neutrons, and electrons directly connects with our periodic table of elements. The atomic number of the elements connects to the number of protons and electrons in the atoms of that element. The amount of protons will always equal the amount of electrons in an atom, the equal but opposite charge of the protons and electrons hold the atom together (electrostatic forces). While the atom is held together due to oppositely charged particles attracting one another, there are also repulsion forces acting between similarly charged particles in the atom (proton to proton or electron to electron interaction). In order to find the number of neutrons in the atoms of a particular element, we must round the atomic mass to the nearest whole number and subtract the atomic number. Found in the nucleus of the atom, the neutrons are neutral in charge so those particles are not influenced by the positive charge of the proton or the negative charge of the electrons.
Practice Problems
|
| ||||||||||||